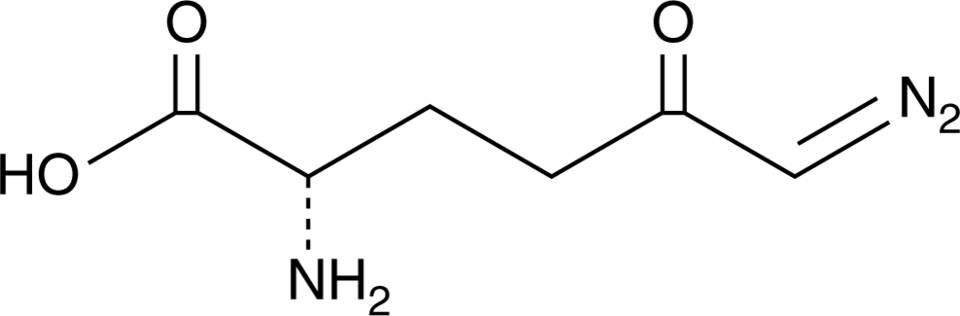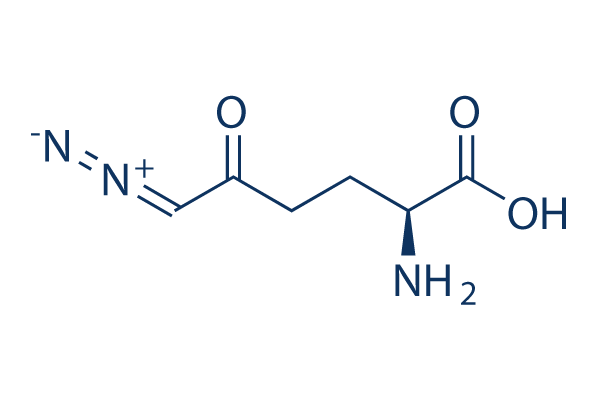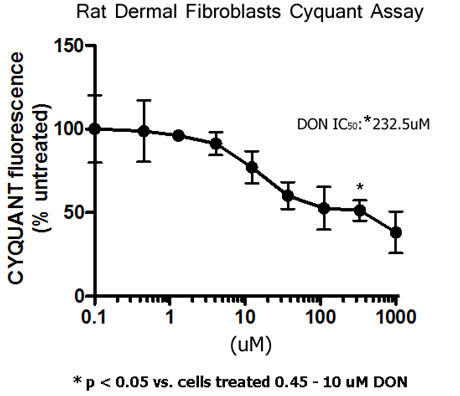
Evaluating the toxicity of the analgesic glutaminase inhibitor 6-diazo-5-oxo -l-norleucine in vitro and on rat dermal skin fibroblasts - MedCrave online
Glutamine deficiency induces DNA alkylation damage and sensitizes cancer cells to alkylating agents through inhibition of ALKBH enzymes | PLOS Biology

Chemical reactivity of 6-diazo-5-oxo-l-norleucine (DON) catalyzed by metalloporphyrins (Fe,Ru) - ScienceDirect

A mechanism behind the antitumour effect of 6-diazo-5-oxo-l-norleucine (DON): disruption of mitochondria - European Journal of Cancer

Model studies towards prodrugs of the glutamine antagonist 6-diazo-5-oxo-l- norleucine (DON) containing a diazo precursor - ScienceDirect
PDF) Discovery of 6-Diazo-5-oxo- l -norleucine (DON) Prodrugs with Enhanced CSF Delivery in Monkeys: A Potential Treatment for Glioblastoma

Tumor-Targeted Delivery of 6-Diazo-5-oxo-l-norleucine (DON) Using Substituted Acetylated Lysine Prodrugs | Journal of Medicinal Chemistry
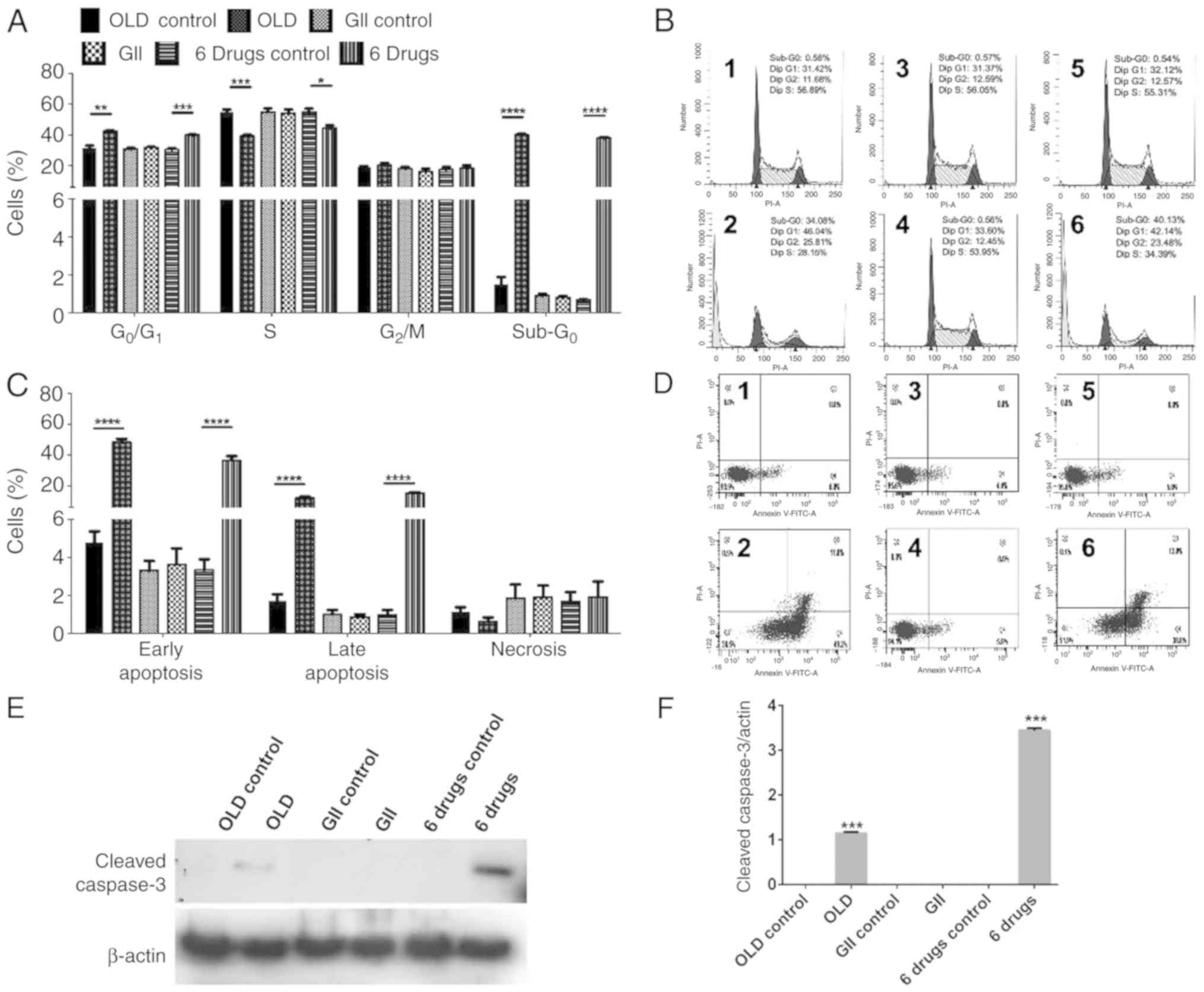
The combination of orlistat, lonidamine and 6‑diazo‑5‑oxo‑L‑norleucine induces a quiescent energetic phenotype and limits substrate flexibility in colon cancer cells
10:6%3C1138::aid-cncr2820100608%3E3.0.co;2-k.fp.png)
Pharmacological and initial therapeutic observations on 6‐Diazo‐5‐Oxo‐L‐ Norleucine (Don) in human neoplastic disease - Magill - 1957 - Cancer - Wiley Online Library

Discovery of 6-Diazo-5-oxo-l-norleucine (DON) Prodrugs with Enhanced CSF Delivery in Monkeys: A Potential Treatment for Glioblastoma | Journal of Medicinal Chemistry
N-(Pivaloyloxy)alkoxy-carbonyl Prodrugs of the Glutamine Antagonist 6-Diazo- 5-oxo-l-norleucine (DON) as a Potential Treatment fo
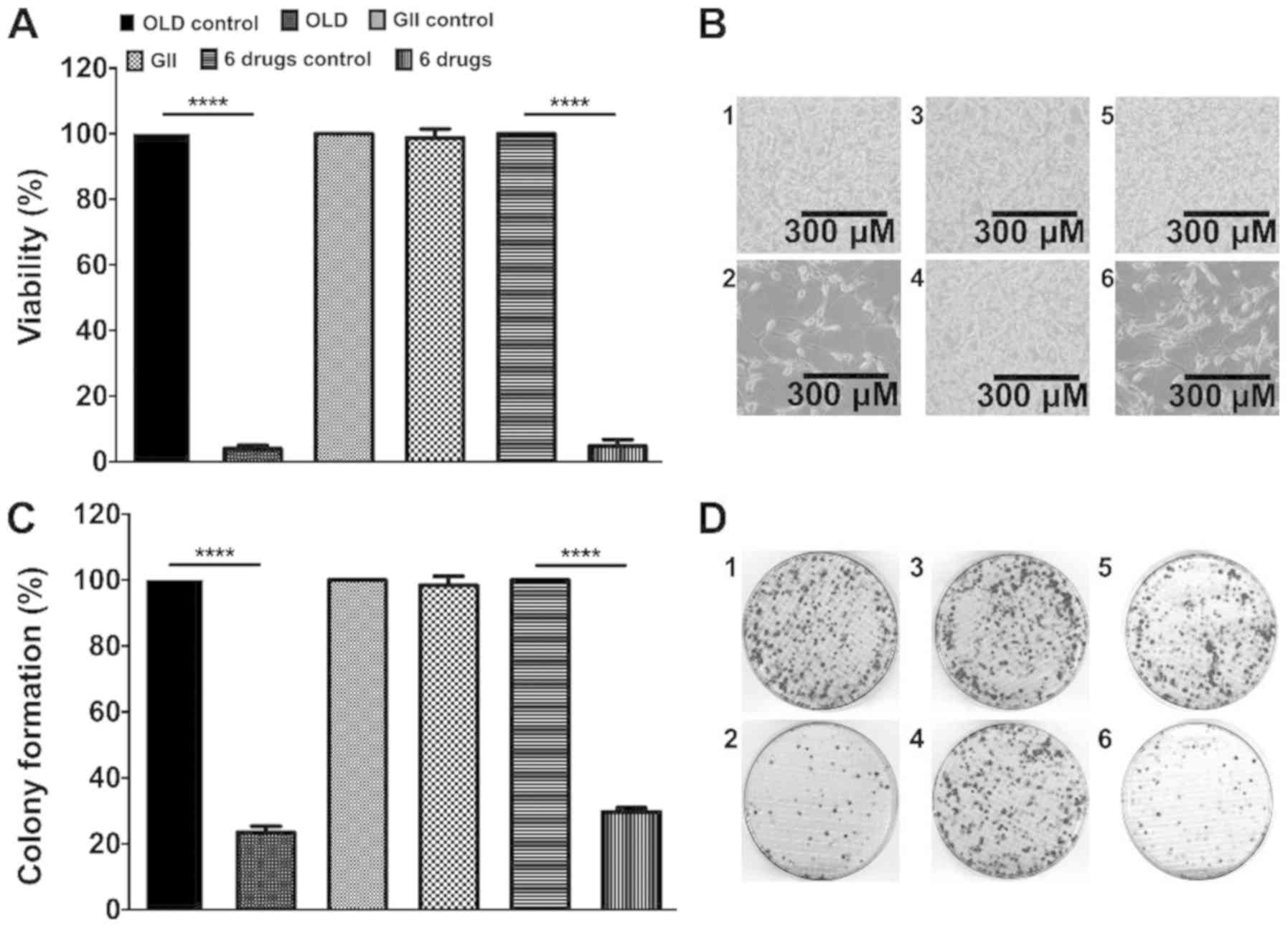
The combination of orlistat, lonidamine and 6‑diazo‑5‑oxo‑L‑norleucine induces a quiescent energetic phenotype and limits substrate flexibility in colon cancer cells